
Covid-19 vaccine second-dose completion and interval between first and second doses among vaccinated persons - United States, December 14, 2020−February 14, 2021. Most vulnerable offered second dose of COVID-19 vaccine earlier to help protect against variants. To use the tool, two inputs are required. The tool below provides guidance on timing for requests to help organize your survey submissions. The company is currently studying JNJ-78436735 as both a two-dose and a one-dose regimen. With informed consent, individuals can choose between a second dose of AstraZeneca or an mRNA vaccine, at an eight- to 12-week interval, recognizing that while waiting 12 weeks helps to. The interval between first and second doses of the Pfizer and Moderna Covid-19 vaccines may be as long as eight weeks for certain people, the US Centers for Disease Control and Prevention said. COVID-19 vaccines that require 2 shots.ĭepartment of Health and Social Care. Plan to administer 2nd dose starting Notification of order will be received by Your vaccines will be delivered between Second Dose Information First Dose Information Moderna Order Request. Second dose of COVID vaccine from Pfizer or Moderna is needed, but timing doesnt have to be exact. Levels and functionality of antibodies after pneumococcal conjugate vaccine in schedules with different timing of the booster dose. I took my second dose of birth control pills earlier than the recommended time, what should I do I took my first dose of levonorgestrel + ethinyl estradiol at 7:20 PM and took my second dose at 7:20 AM by mistake as I have thought that the interval was 12 hours, when the actual recommended interval was 24 hours. 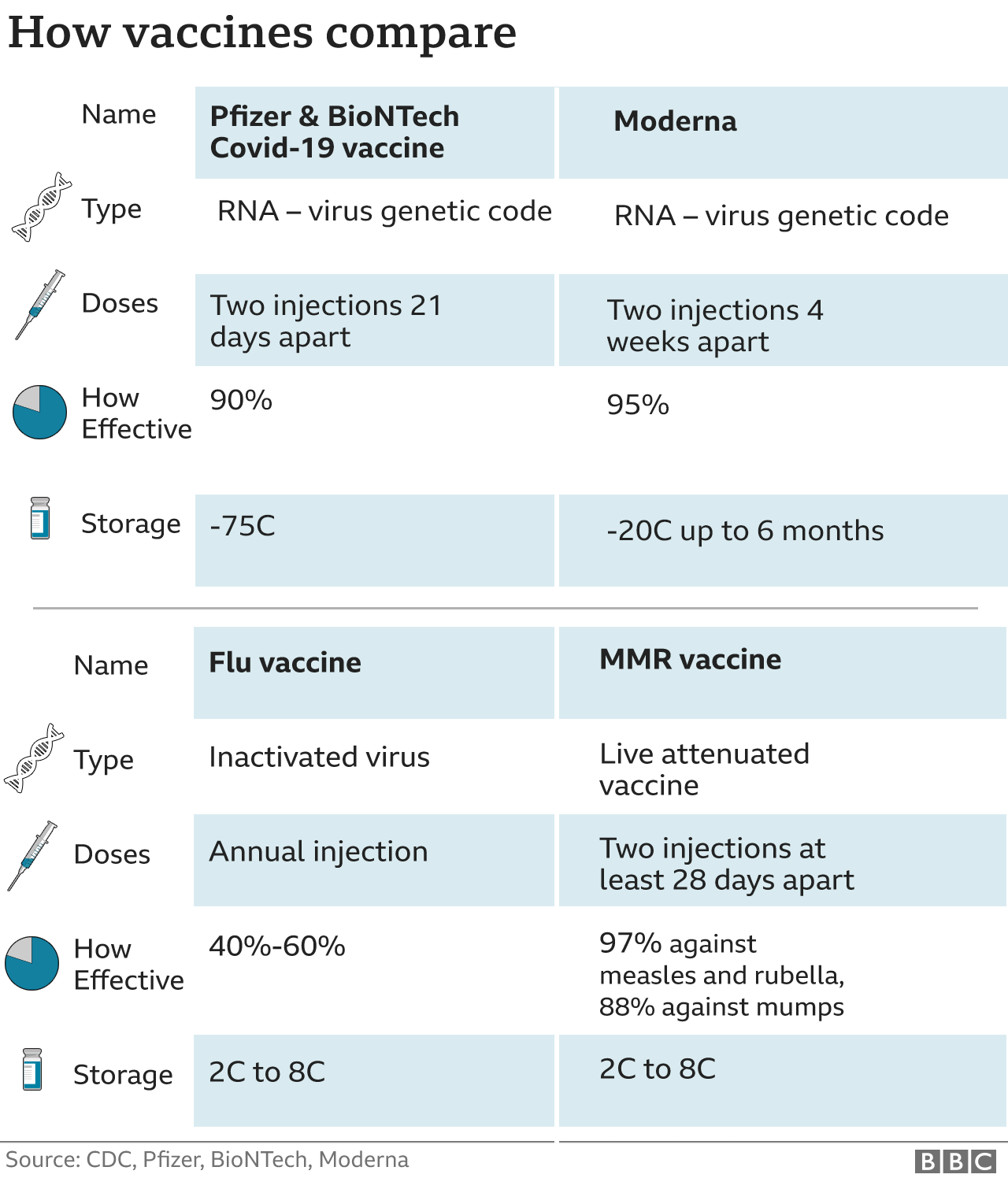 
Van Westen E, Rodenburg GD, van Gils EJM, et al. Nevertheless, extensive data on the immune responses of hematological patients and the efficacy of the third dose of the vaccine are scarce. The recommended interval between the first and second dose is 21 days for Pfizer-BioNTech and 28 days for Moderna in this study, second doses received 1725 days (Pfizer-BioNTech) and 2432 days (Moderna) after the first dose were included. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials. Patients with B-cell malignancies are at a higher risk of severe SARS-CoV-2 infections. Voysey M, Costa Clemens SA, Madhi SA, et al. Extended interval BNT162b2 vaccination enhances peak antibody generation in older people.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
